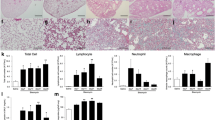
Summary
Continuous beta blockade stimulates deposition of collagen in the pulmonary alveolar interstitium of adult rats. It also causes changes to the capillary endothelial cell compartment reminiscent of programmed cell death. To test whether beta blockade results in endothelial cell apoptosis, cultures of capillary endothelial cells were treated with both a wide-spectrum beta blocker and a beta-2-specific antagonist. Apoptosis was measured in these cultures using both terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling and annexin-V assays. Both forms of beta blockade stimulated programmed cell death in these cultures. To test whether the apoptotic effect of beta blockade was related to interstitial collagen deposition, capillary endothelial cells were cocultured with beta-blocked pulmonary fibroblast monolayers. Cocultured endothelial cells were substantially protected from apoptosis after beta blockade; coculture over plain tissue culture plastic or over exogenous collagen films had no effect on programmed cell death in endothelial cells. These results suggest that both pulmonary endothelial and interstitial cells are vulnerable to injury from beta blockade but that paracrine interactions between these cells may protect the peripheral lung from substantive damage.
Similar content being viewed by others
References
Adamson, I. Y. Drug-induced pulmonary fibrosis. Environ. Health Perspect. 55:25–36; 1984.
Ahmad, S.; Chrétien, P.; Daniel, E. E.; Shen, S. H. Characterization of beta adrenoceptors on cultured endothelial cells by radioligand binding. Life Sci. 47:2365–2370; 1990.
Bachetti, T.; Morbidelli, L. Endothelial cells in culture: a model for studying vascular functions. Pharmacol. Res. 42:9–19; 2000.
Bruce, M. C.; Honaker, C. E.; Cross, R. J. Lung fibroblasts undergo apoptosis following alveolarization. Am. J. Respir. Cell Mol. Biol. 20:228–236; 1999.
Carrington, C. B. Organizing interstitial pneumonia: definition of the lesion and attempts to devise an experimental model. Yale J. Biol. Med. 40:352–363; 1968.
Chen, B.; Henke, C.; Snover, D.; Wendt, D.; Ingbar, D.; Bitterman, P. Role of mesenchymal cell death in lung remodeling after injury. J Clin. Invest. 92:388–397; 1993.
Choi, K. B.; Wong, F.; Harlan, J. M.; Chaudhary, P. M.; Hood, L.; Karsan, A. Lipopolysaccharide mediates endothelial apoptosis by a FADD-dependent pathway. J. Biol. Chem. 273(32):20185–20188; 1998.
Crystal, R. J.; Bitterman, P.; Rennard, S.; Hance, A.; Keogh, B. Interstitial lung disease of unknown cause (second of two parts). New Engl. J. Med. 310:235–244; 1984.
Crystal, R. J.; Gadek, J.; Ferrand, V.; Fulmer, J.; Line, B.; Hunninghake, G. Interstitial lung disease: current concepts of pathogenesis, staging and therapy. Am. J. Med. 70:542–568; 1981.
Dunsmore, S. E.; Rannels, D. E. Extracellular matrix biology in the lung. Am. J. Physiol. Lung Cell Mol. Physiol. 270:L3-L27; 1996.
Flavin, M. P.; Parton, L. A.; Bowman, C. M. Conditioned medium from irradiated bovine pulmonary artery endothelial cells stimulates increased protein synthesis by irradiated bovine lung fibroblasts in vitro. Exp. Lung Res. 16(5):405–422; 1990.
Fujita, M.; Kuwano, K.; Kunitake, R.; Hagimoto, N.; Miyazaki, H.; Kaneko, Y.; Maeyama, T.; Hara, N. Endothelial cell apoptosis in lipopolysaccharide-induced lung injury in mice. Int. Arch. Allergy Immunol. 117:202–208; 1998.
Gerschenson, L. E.; Rotello, R. J. Apoptosis: a different kind of cell death. FASEB J. 6:2450–2455; 1992.
Hoyt, D. G.; Mannix, R. J.; Rusnak, J. M.; Pit, B. R.; Lazo, J. S. Collagen is a survival factor against LPS-induced apoptosis in cultured sheep pulmonary artery endothelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 269:L171-L177; 1995.
Kockx, M. M.; Knaapen, M. W. M. The role of apoptosis in vascular disease. J. Pathol. 190:267–280; 2000.
Lossos, I. S.; Reuven, O.; Goldstein, R. H.; Conner, M. W.; Breuer, R. Amelioration of bleomycin-induced pulmonary injury by cyclosporin A. Exp. Lung Res. 10:331–355; 1995.
Maulik, N.; Kagan, V. E.; Tyurin, V. A.; Das, D. K. Redistribution of phosphatidylethanolamine and phosphatidylserine precedes reperfusion-induced apoptosis. Am. J. Physiol. Heart Circ. Physiol. 274(43):H242-H248; 1998.
Reiser, K.; Last, J. Early cellular events in pulmonary fibrosis. Exp. Lung Res. 10:331–355; 1986.
Sano, M.; Yoshimasa, T.; Yagura, T.; Yamamoto, I. Non-homogeneous distribution of beta-1 and beta-2-adrenceptors in various human tissues. Life Sci. 52:1063–1070; 1993.
Schumann, R. R.; Belka, C.; Reuter, D.; Lamping, N.; Kirschning, C. J.; Weber, J. R.; Pfeil, D. Lipopolysaccharide activates caspase-1 (interleukin-1-converting enzyme) in cultured monocytic and endothelial cells. Blood 91(2):577–584; 1998.
Schwartz, M. The acute (noninfectious) interstitial lung diseases. Compr. Ther. 22:622–630; 1996.
Smith, D. M.; Sommers Smith, S. K. The fibrogenic response of adult rat lung to continuous propranolol treatment. Histol. Histopathol. 3:291–299; 1988.
Sommers Smith, S. K.; Giannopoulos, G. Identification of beta-adrenergic receptors in isolated pulmonary alveolar type II cells from fetal and adult rabbits. Life Sci. 33:2071–2076; 1983.
Sommers Smith, S. K.; Smith, D. M. Effect of continuous beta-blockade on collagen synthesis in interstitial fibroblasts isolated from adult rat lung. An in vitro model of progressive pulmonary fibrosis. In Vitro Toxicol. 3:129–136; 1989.
Sommers Smith, S. K.; Smith, D. M. Stimulation of collagen synthesis and growth by propranolol and by beta-blocked alveolar macrophages. In Vitro Toxicol. 10:233–244; 1997.
Sulkowska, M.; Sulkowska, S. Apoptosis-like changes in the lungs induced by cyclophosphamide and papain. I. An ultrastructural study. J. Submicros. Cytol. Pathol. 30:105–116; 1998.
Uhal, B.; Iravati, J.; True, A.; Mundle, S.; Raza, A.; Pardo, A.; Selman, M. Fibroblasts isolated after fibrotic lung injury induce apoptosis of alveolar epithelial cells in vivo. Am. J. Physiol. Lung Cell Mol. Physiol. 308:L819-L828; 1995.
Wendt, C. H.; Polunovsky, V. A.; Peterson, M. S.; Bitterman, P. B.; Ingbar, D. H. Alveolar epithelial cells regulate the induction of endothelial cell apoptosis. Am. J. Physiol. 267:C893-C900; 1994.
Wu, C.; Bishopric, N.; Pratt, R. Atrial naturetic peptide induces apoptosis in neonatal rat cardial myocytes. J. Biol. Chem. 272(24):14860–14866; 1997.
Xia, Z.; Dickens, M.; Raingeaud, J.; Davis, K.; Greenberg, M. Opposing effects of ERK and TNK-p38 MAP kinases on apoptosis. Science 270:1326–1331; 1995.
Zoellner, H.; Höfler, H. M.; Beckman, R., et al. Serum albumin is a specific inhibitor of apoptosis in human endothelial cells. J. Cell Sci. 109:2571–2580; 1996.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sommers Smith, S.K., Smith, D.M. Beta blockade induces apoptosis in cultured capillary endothelial cells. In Vitro Cell.Dev.Biol.-Animal 38, 298–304 (2002). https://doi.org/10.1290/1071-2690(2002)038<0298:BBIAIC>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2002)038<0298:BBIAIC>2.0.CO;2